Løste krise med eget produkt
Tekst: Anne-Lise Aakervik
Foto: Synlig.no v/Erik Børseth
Da produsenten brått trakk tilbake et viktig medisinsk nett som brukes for å holde stomibrokk på plass, måtte legene selv trå til med nål og tråd.
.

Fra høyre kirurgene Brynjulf Ystgaard og Tor Eivind Bernstein, samt Tonje S.Steigedal fra TTO. Kirurg Tommy Bernhard Johnsen har også jobbet med oppfinnelsen, men var ikke tilstede da bildet ble tatt.
Dette var et av få nett på markedet som var egnet til formålet. Men gastrokirurgene ved St. Olavs Hospital var ikke rådville. De tok bokstavelig talt synålen i egne hender og løste krisa med et forbedret nett. Nå søker de patent på løsningen og vil gjerne få det distribuert ut i verden.
– Det var faktisk ganske kritisk for oss, sier kirurg Tor Eivind Bernstein som er en av de tre oppfinnerne. Han er tillegg til å være lege ved St. Olavs Hospital også knyttet til institutt for klinisk og molekylær medisin ved NTNU.
– Vi hadde flere operasjoner planlagt da dette nettet plutselig ble trukket fra markedet. Årsaken var at sømmene på nettet viste seg å være svake. De kunne ganske enkelt rakne og derfor ble produktet trukket fra markedet over hele verden.
Igjen sto kirurgene og pasientene med en annen type nett laget av svinebindevev, men dette er ikke så holdbart som det andre og få bruker det i permanente løsninger.
Uten alternativer var gode råd dyre. – Det første vi gjorde var å sjekke litt rundt for å høre hva andre sykehus gjorde, men noen løsning fant vi ikke. Da bestemte vi oss for å gjøre det selv, og løsningen har vist seg å fungere bra. Så bra at vi håper det er noen som kan tenke seg å lisensiere produktet.
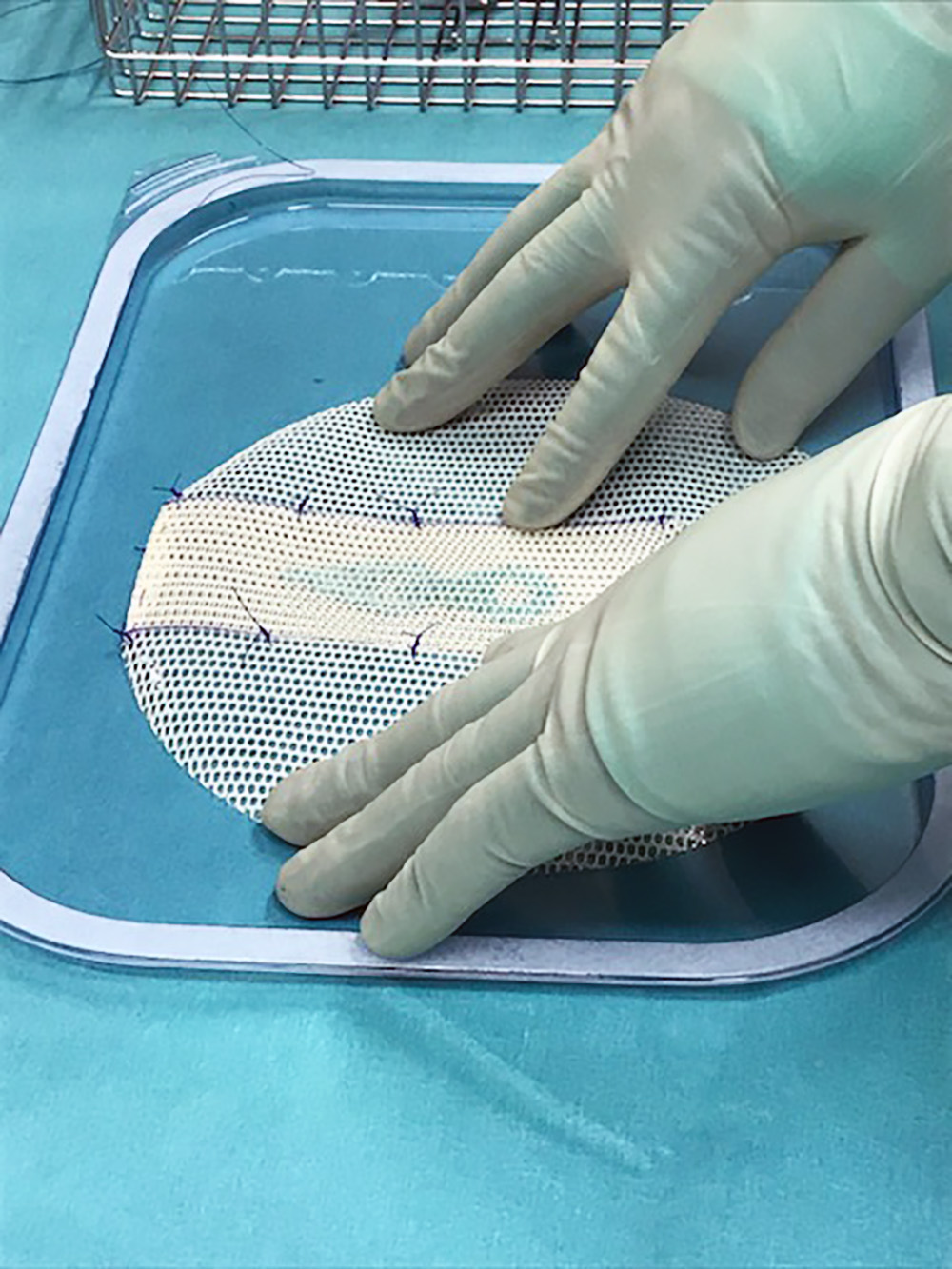
Satte seg med nål og tråd: Da et kirurgisk nett som brukes ved stomioperasjoner ble trukket fra markedet måtte kirurgene finne egen løsning. Det ble «Bakermesh», et forbedret og tilpasset nett.
Stort behov
Pasientene har behov for slike nett er de som har fått utlagt tarm eller urinrør, såkalt stomi. På verdensbasis er det 15 millioner, og omtrent halvparten vil få brokk etter operasjonen hvor man etablerer den utlagte tarmen/urinrøret. Brokk defineres som en utposing av bukhulen gjennom et svekket område i muskelveggen. Dette gjør gjerne at stomiposen mister festet og det må en ny operasjon til for å holde brokken på plass. Dette er oppgaven til nettet som plasseres mot bukveggen for å styrke den.
Nettet som ble trukket brukes i en prosedyre som kalles sandwich ettersom det legges sammen med en annen type nett som en sandwich, og er blitt brukt i mange år. Helt til det altså tok slutt i oktober i fjor. Det er et syntetisk nett som har vist seg å fungere veldig godt.
Krise skaper kreativitet
Gastrokirurgene kan fra før håndtere en synål, også i andre materialer enn menneskevev. – Kostnaden på slike nett er ekstremt høy, så vi kunne ikke bruke det samme materialet i designfasen, men så snart vi hadde et nett vi visste ville fungere, ble det laget i riktig materiale og testet under operasjoner siden oktober i 2018.
– Vi følger opp denne gruppen med pasienter tett. Det er endelige stomibrukere som ikke skal operere tarmen tilbake igjen som får disse nettene, forklarer Bernstein. –Resultatene har vært gode. Vi har dokumentert at dette fungerer godt, både under, etter og på sikt. Det har ikke vært noen uregelmessigheter etter at vi har operert inn vår løsning. Og nå har vi fulgt de første pasientene over seks måneder og planlegger ett års kontroll.

Patentsøknad
Sammen med Tonje S. Steigedal fra TTO har de jobbet med patentsøknad har presentert oppfinnelsen for potensielle samarbeidspartnere fra industrien. De håper at produktet de har utviklet kan utlisensieres til en produsent slik at de slipper å lage dem selv etter hvert som behovet oppstår.
– Vi har jo ikke laget noe revolusjonerende nytt, det er vanskelig, men vi har gjort viktige tilpassinger slik at man ikke skal oppleve at sømmene i nettet rakner f.eks.
Hvis ingen ønsker å produsere dette må legene selv lage disse nettene etter hvert som behovet oppstår. Da bruker de rundt 10-15 minutter ekstra under operasjonen til å sy sammen et nett, da de ikke kan drive forhåndsproduksjon. – Vi ønsker selvsagt et ferdigprodusert nett som vi kan ta ut av pakningen.
– Vi vet at det er et behov som er nødvendig ved denne typen operasjoner, derfor tror vi at det er gode muligheter for å få en industriaktør til å bistå med regulatorisk godkjenningsprosess og kommersialisering av det nye nettet. Industriaktørene uttrykker interesse for produktet og ønsker å følge tett med på resultatene fra den kliniske utprøvingen.
Med midler fra NTNU Discovery har de ferdigstilt patentsøknaden og utformet markedsføringspakke.
Prosjekter og nyheter
Kontakt:
Prosjektleder
Jan Hassel
Epost: jan.hassel@ntnu.no
Telefon: 906 53 180
Kontor: Hovedbygget, sokkel
Håvard Wibe
Epost: havard.wibe@ntnu.no
Telefon: 41 47 37 68
Kontor: Hovedbygget, sokkel







